ENCAB000AAT
Antibody against Homo sapiens CREM
Homo sapiens
K562, GM12878, HepG2, MCF-7
characterized to standards with exemption
- Status
- released
- Source (vendor)
- Sigma
- Product ID
- WH0001390M2
- Lot ID
- 11056-3B5
- Characterized targets
- CREM (Homo sapiens)
- Host
- mouse
- Clonality
- monoclonal
- Isotype
- IgG1κ
- Antigen description
- CREM (NP_853549, a.a. 201-301) partial recombinant protein with GST tag.
- External resources
Characterizations
CREM (Homo sapiens)
compliant
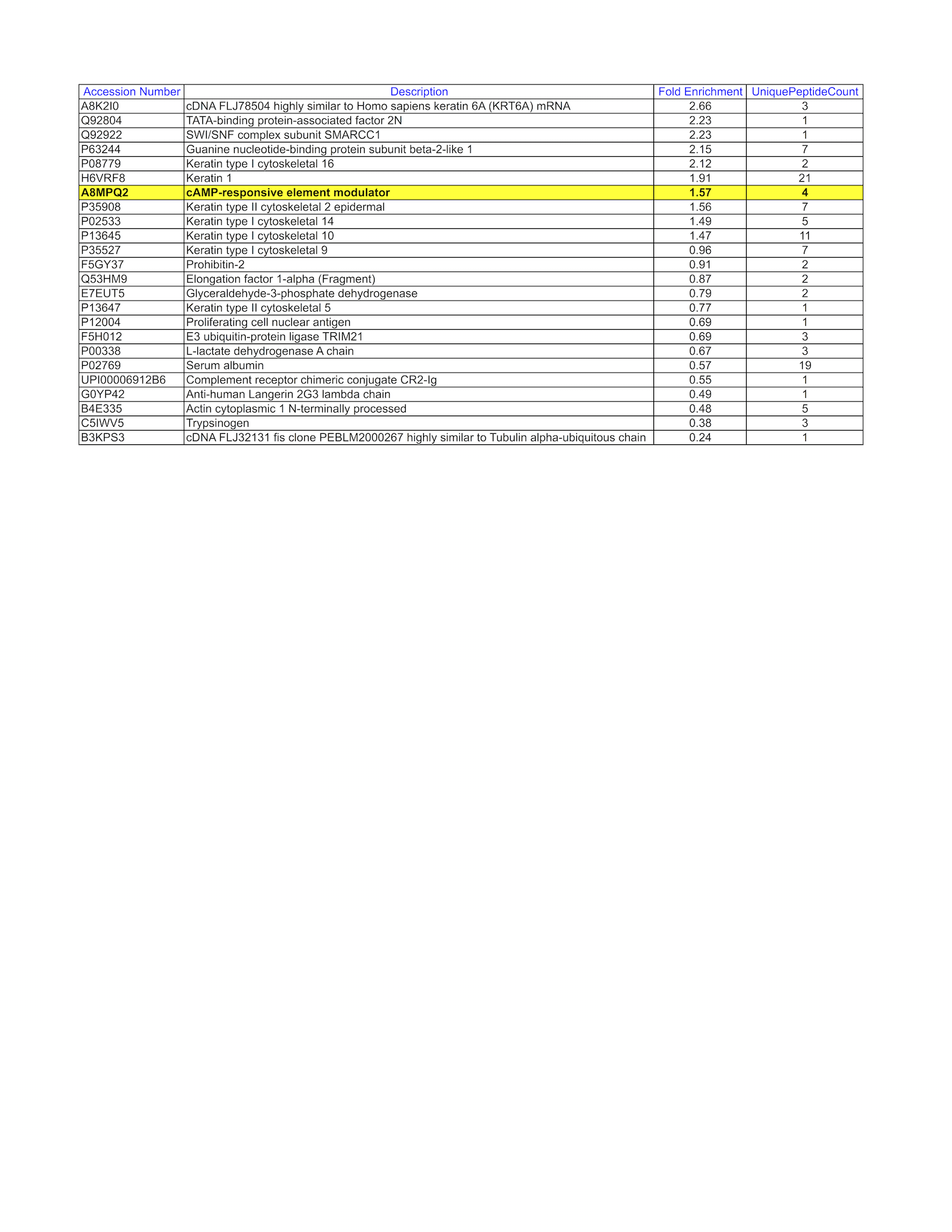
- Caption
- Analysis of gel fragment 1 from GM12878: The sample was analyzed on a LTQ XL Linear Ion Trap Mass Spectrometer by LC-ESI-MS/MS. Peptides were identified using SEQUEST tandem mass spectral analysis with probability based matching at p < 0.05. SEQUEST results were reported with ProteinProphet protXML Viewer (TPP v4.4 JETSTREAM) and filtered for a minimum probability of 0.9. All protein hits that met these criteria were reported, including common contaminants. Fold enrichment for each protein reported was determined using a custom script based on the FC-B score calculation from the reference Mellacheruvu et al., 2013. The CRAPome: a contaminant repository for affinity purification mass spectrometry data. Nat. Methods. 10(8):730-736. Doi:10.1038/nmeth.2557. The target protein, CREM, was identified as the 7th ranked enriched protein and the 2nd ranked transcription factor based on IP-Mass Spectrometry.
- Submitted by
- Collin White
- Lab
- Richard Myers, HAIB
- Grant
- U54HG006998
- Download
- CREM-1_ENCAB000AAT.png
CREM (Homo sapiens)
compliant
- Caption
- Analysis of gel fragment 2 from GM12878: The sample was analyzed on a LTQ XL Linear Ion Trap Mass Spectrometer by LC-ESI-MS/MS. Peptides were identified using SEQUEST tandem mass spectral analysis with probability based matching at p < 0.05. SEQUEST results were reported with ProteinProphet protXML Viewer (TPP v4.4 JETSTREAM) and filtered for a minimum probability of 0.9. All protein hits that met these criteria were reported, including common contaminants. Fold enrichment for each protein reported was determined using a custom script based on the FC-B score calculation from the reference Mellacheruvu et al., 2013. The CRAPome: a contaminant repository for affinity purification mass spectrometry data. Nat. Methods. 10(8):730-736. Doi:10.1038/nmeth.2557. The target protein, CREM, was identified as the 1st ranked enriched protein and the 1st ranked transcription factor based on IP-Mass Spectrometry.
- Submitted by
- Collin White
- Lab
- Richard Myers, HAIB
- Grant
- U54HG006998
- Download
- CREM-2_ENCAB000AAT.png
CREM (Homo sapiens)
K562GM12878
exempt from standards
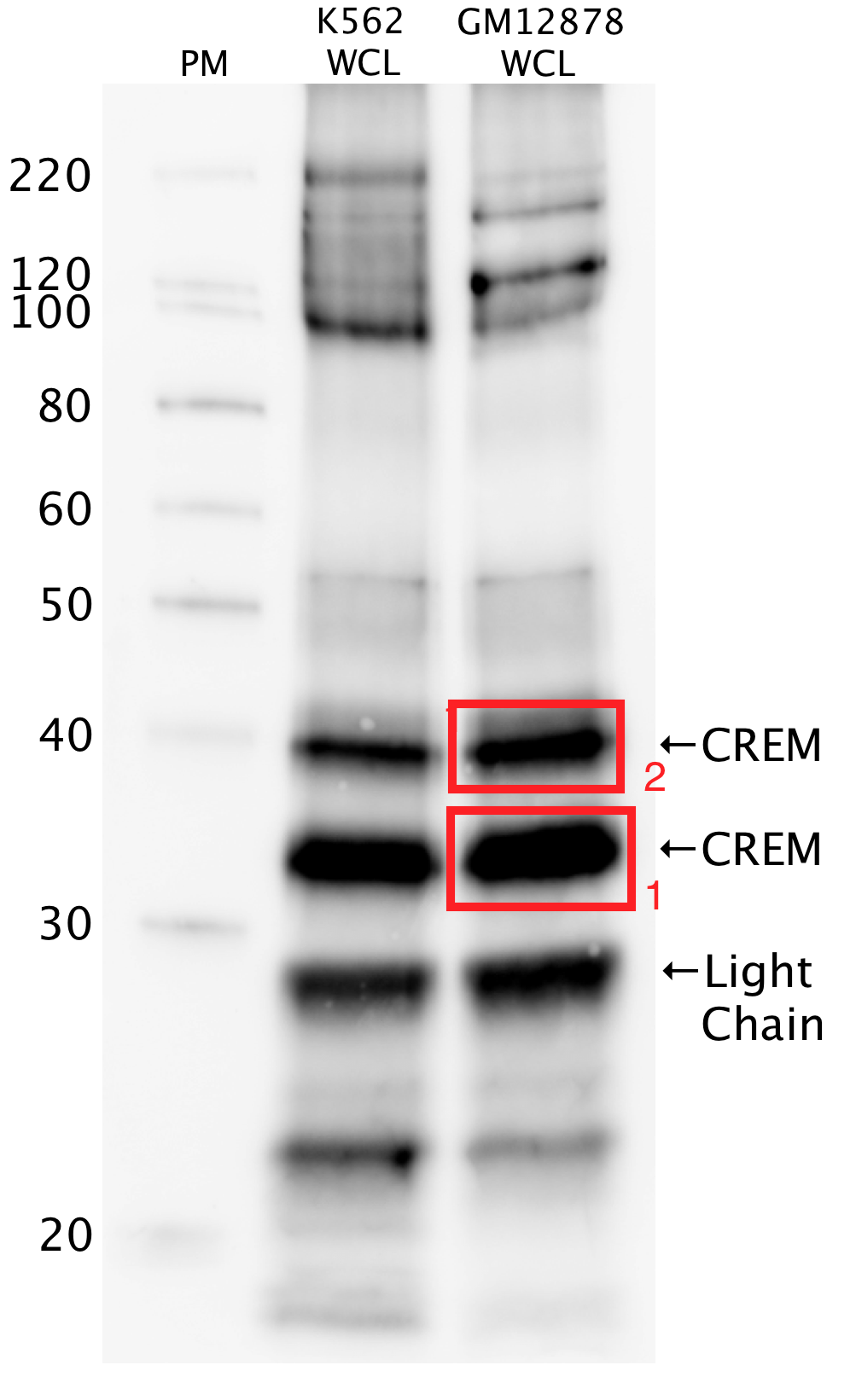
- Caption
- Whole cell lysates of K562 and GM12878 were immunoprecipitated using the primary antibody (Sigma; WH0001390M2). The IP fraction was separated on a 12% acrylamide gel with the Bio-Rad PROTEAN II xi system. After separation, the samples were transferred to a nitrocellulose membrane with an Invitrogen iBlot system. The membrane was probed with the primary antibody (same as that used for IP) and a secondary HRP-conjugated antibody. The resulting bands were visualized with SuperSignal West Femto solution (Thermo Scienfiic). Protein marker (PM) is labeled in kDa. Two bands were detected at ~35 and ~40kDa.
- Submitter comment
- These do not have bands at the expected sizes, but are exempted by having mass spectrometry analysis on both bands.
- Reviewer comment
- This antibody has a passing mass spectrometry characterization.
- Submitted by
- Flo Pauli-Behn
- Lab
- Richard Myers, HAIB
- Grant
- U54HG006998
- Download
- CREM_IP-MS.png
CREM (Homo sapiens)
K562GM12878HepG2MCF-7
exempt from standards
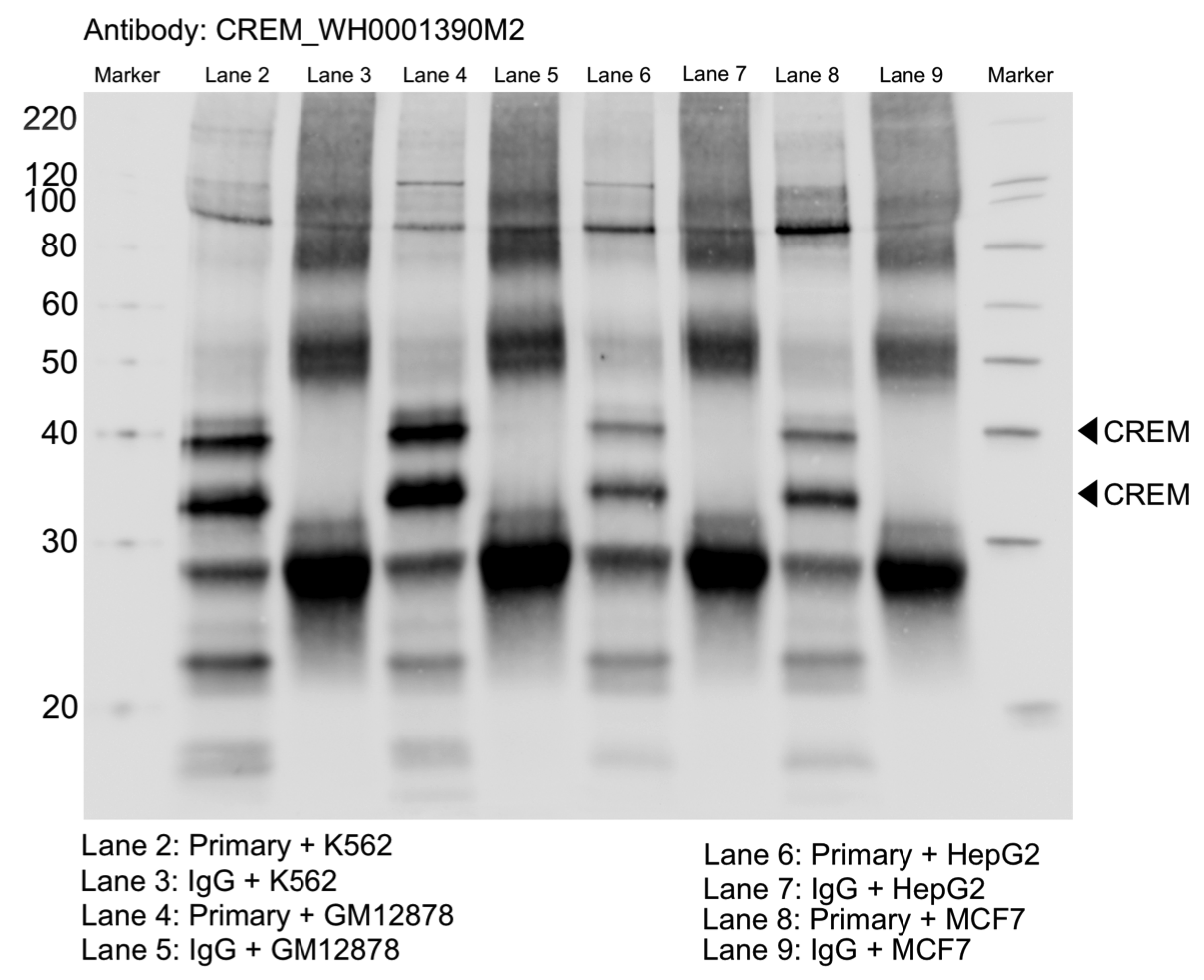
- Caption
- Whole cell lysates of K562, MCF7, GM12878 and HepG2 were immunoprecipitated using the primary antibody (Sigma; WH0001390M2). The IP fraction was separated on a 12% acrylamide gel with the Bio-Rad PROTEAN II xi system. After separation, the samples were transferred to a nitrocellulose membrane with an Invitrogen iBlot system. The membrane was probed with the primary antibody (same as that used for IP) and a secondary HRP-conjugated antibody. The resulting bands were visualized with SuperSignal West Femto Solution (Thermo Scientific). Protein Marker (PM) is labeled in kDa. Two bands were detected at ~34 and 39 kDa.
- Submitter comment
- --
- Reviewer comment
- Band of interest is not 50% of the overall signal in the lane
- Submitted by
- Mark Mackiewicz
- Lab
- Richard Myers, HAIB
- Grant
- U54HG006998
- Download
- CREM_IP-WB2.png
CREM (Homo sapiens)
K562HepG2
not reviewed
- Caption
- GM12878 whole cell lysate was immunoprecipitated using the primary antibody (Sigma; WH0001390M2). The IP fraction was loaded on a 12% Bio-Rad TGX gel and separated with the Bio-Rad Tetra Cell System. Gel fragments (rectangle outline) corresponding to the bands indicated on the Western blot image were excised and sent to the University of Alabama at Birmingham Cancer Center Mass Spectrometry/Proteomics Shared Facility.
- Submitter comment
- Non specific bands and no IgG control, but are exempted due to mass spec analysis
- Reviewer comment
- non specific bands and no IgG control
- Submitted by
- Flo Pauli-Behn
- Lab
- Richard Myers, HAIB
- Grant
- U54HG006998
- Download
- CREM_IP_WB_MS_1.png